Power from Zinc and Copper in Action - News about Energy Storage, Batteries, Climate Change and the Environment

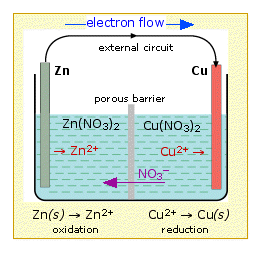
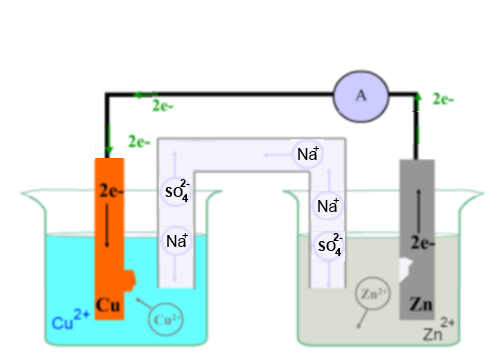
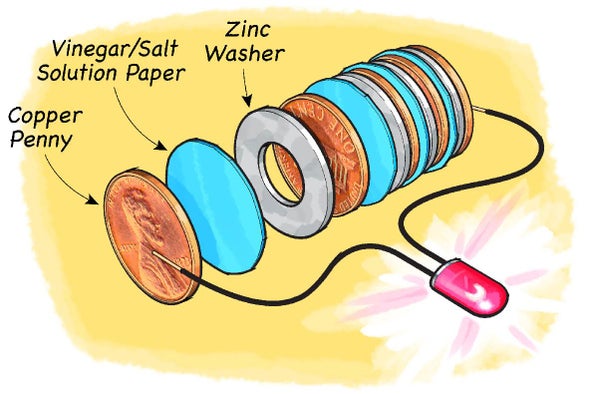
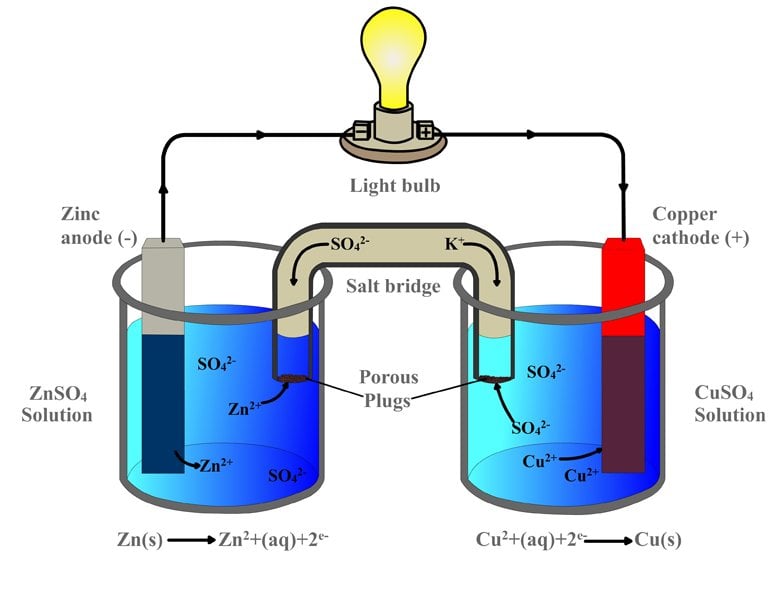
Simple Voltaic Cells batteries of copper zinc magnesium dipped in salt solution electrolyte investigating reactivity series of metals using simple cell and voltmeter gcse chemistry KS4 science igcse O level revision notes

Simple Voltaic Cells batteries of copper zinc magnesium dipped in salt solution electrolyte investigating reactivity series of metals using simple cell and voltmeter gcse chemistry KS4 science igcse O level revision notes

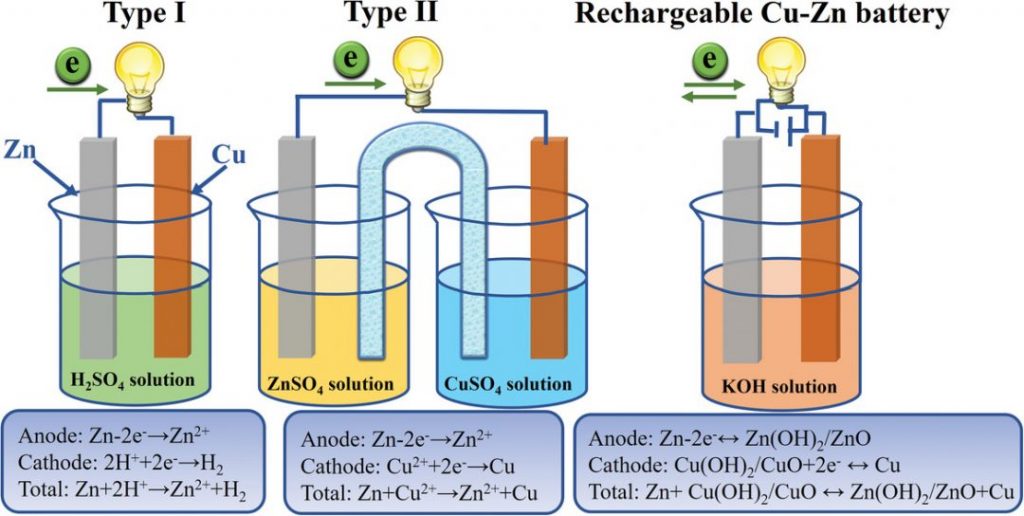
Figure 13. Photos of the used copper and zinc sheet. Top: before, bottom: after use as electrodes in the Cu-Zn battery. On top: white: the separators. The materials were winded as a
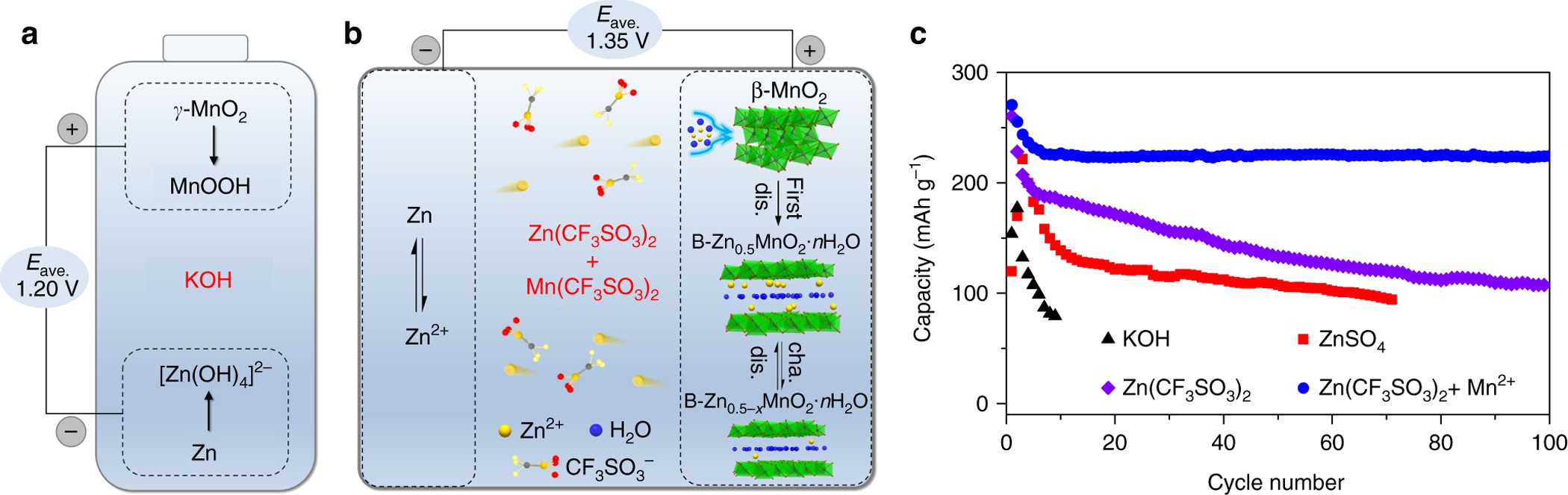
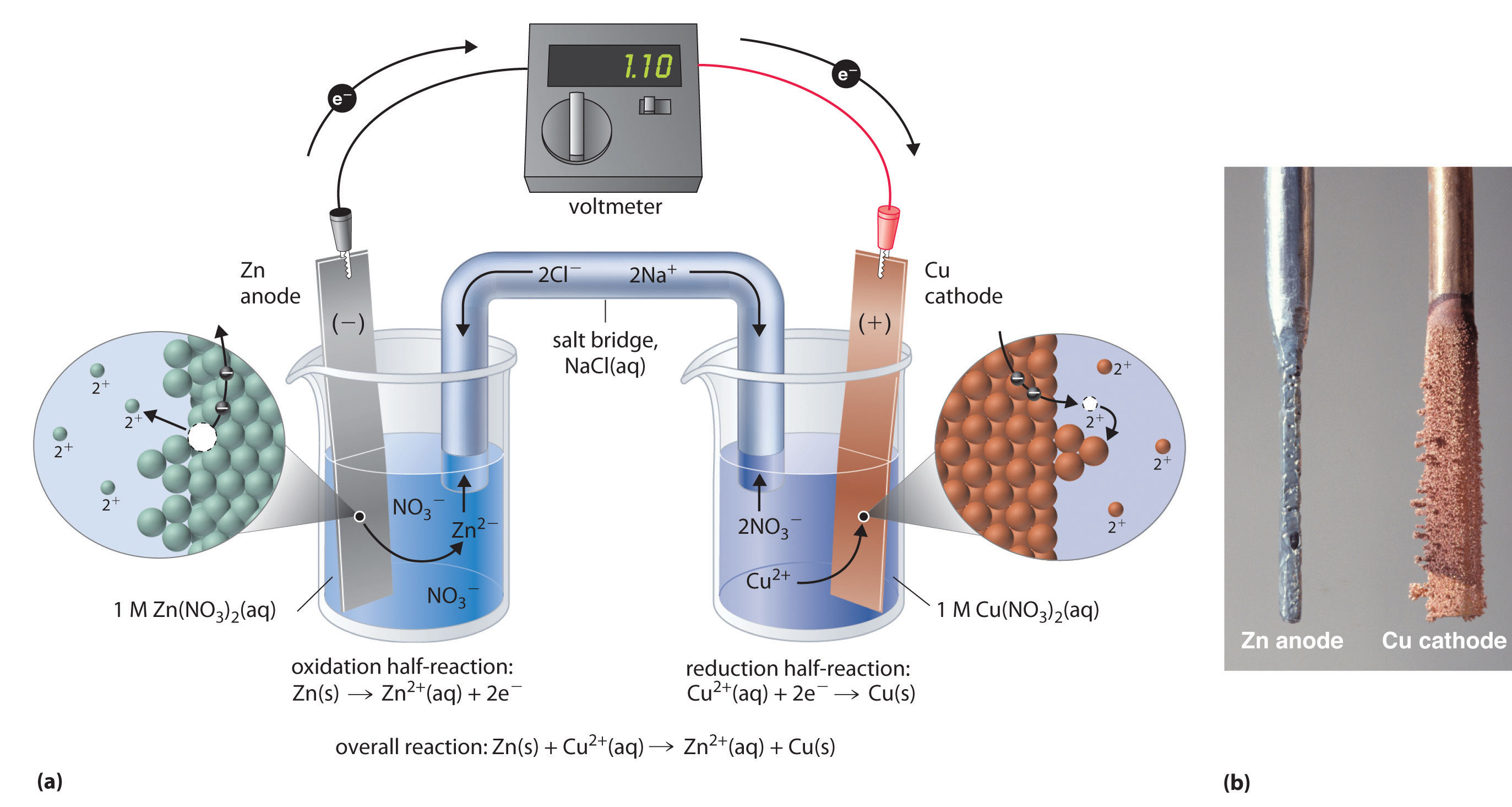
Rechargeable aqueous zinc-manganese dioxide batteries with high energy and power densities | Nature Communications

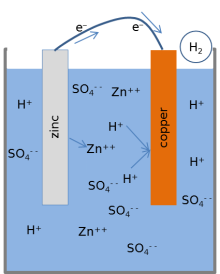
9 Diagram of a simple zinc-copper battery. Because zinc sulfate is more... | Download Scientific Diagram